Installation
Med nanos are injected into any major blood vessel. Within the bloodstream, they swim to various attachment points according to onboard AI logic and commands from the master assembly bots.
Like sea turtles hatching from their eggs on the shore, some nano bots won’t make it to their destinations. Some become defective. Others might contain burn-in defects from the fabrication process that don’t appear until after a few days of use. So, redundancy is key.

But redundancy must be managed. Especially if two chipsets are mapping data into the same RAM chip at the same time. Each chipset bot locates a processor core bot and RAM bot and tethers itself between them. It establishes control if another bot hasn’t already done so. Otherwise, it waits in case the currently controlling chipset bot fails, is removed by a demo bot, or gets reassigned to another core by the master assembly bots.
A master assembly bot swarm detects overall assembly problems that individual bot AI logic can’t see, and reallocates bots as needed. If it can’t, it reports this to system diagnostics. Doctors see these errors on their HUDs and inject replacements.
Removal
Either because bots are damaged beyond repair or the user wants a newer, flashier model, you sometimes need to remove old med nanos. To do this, the doctor injects a round of demo bots. These guys are programmed to go after the specific bots marked for removal. The demo bots swim to their targets, shutdown their onboard CPUs, detach them (either mechanically or chemically), and turn themselves off.
The demo and target bots get flushed away into the veins and end up in the kidneys to pass with urine. Once the signals from all demo bots have died off, doctors inject replacement bots. Both could happen at the same time, since the demo bots should ignore the new upgrade bots, but it’s safer to keep these as two separate steps.
Anti-Aging
While I don’t have a definitive answer for how med nanos would prevent aging, I have a reasonable guess. It involves protecting telomeres, which are extra nucleotides at the end of each chromosome.
(https://commons.wikimedia.org/wiki/File:Telomeres.png )
As people age, telomeres shorten, which can make cells unable to repair themselves and cause them to self-destruct by apoptosis (programmed cell death) or become senescent (the cell stops dividing). Senescent cells release inflammatory chemicals that tell the immune system to come and remove them. Either way, when telomeres become too short, the cell will die or become cancerous. Or lead to other diseases.
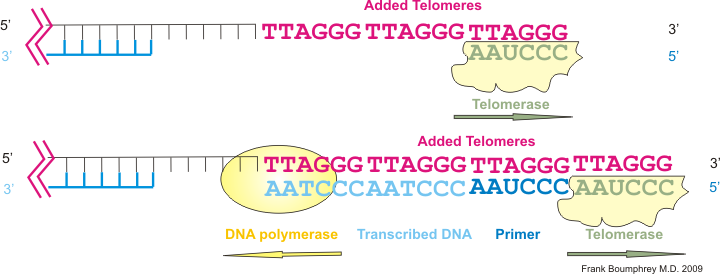
Med nanos to the rescue! They reactivate an enzyme that’s suppressed in normal somatic cells called telomerase, which adds “TTAGGG” repeats to the ends of chromosomes to rebuild telomeres.
(T=thymine, A=adenine, and G=guanine are DNA nucleobases.)
(https://commons.wikimedia.org/wiki/File:Telomerase.png)
As nice as this sounds, using telomerase to preserve telomere length comes with risk. Most cancer cells use this very trick to keep themselves alive. No worries. Med nanos distinguish between cancerous cells and normal cells and kill the cancerous ones before they can metastasize (spread). More often, they detect genetic mutations right when they occur and correct them on-the-spot, preventing cancer cells from forming in the first place!
Sources:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3370421/
https://pubmed.ncbi.nlm.nih.gov/18391173/
https://en.wikipedia.org/wiki/Cellular_senescence
https://longevitylive.com/anti-aging/anti-aging-secrets-can-telomeres-tell/
https://www.faim.org/telomeres-increase-longevity-by-protecting-your-dna
If med nanos became available in real life for human clinical trials, would you sign up to take part? Reply to this email and let me know.
See you next month!
Dylan

Thanks for your blog, nice to read. Do not stop.
Glad you’re enjoying these! I plan to keep them coming every month 🙂